How to find investors for SaMD startup?
As innovation keeps on propelling all aspects of medical services, programming has turned into a significant piece, all things considered, coordinated generally into advanced stages that serve both clinical and non-clinical purposes. Programming, which all alone is a medical device – Software as a Medical Device – is one of three sorts of programming identified with clinical gadgets. The other two kinds of programming identified with medical devices incorporate programming vital to a clinical gadget (Software in a medical device) and programming utilized in the production or upkeep of a clinical gadget.
What is Software as a Medical Device?
The term Software as a Medical Device External Link Disclaimer is characterized by the International Medical Device Regulators Forum (IMDRF) as "programming planned to be utilized for at least one clinical purpose that plays out these reasons without being important for an equipment medical device."

Utilization of Software as a Medical Device is proceeding to increment. The software as a medical device market is expected to reach $86 billion in 2027. Increasing SaMD popularity resides in the capability to facilitate disease diagnosis, management, and treatment using a SaMD on general-purpose deviTo give some examples, its. It tends to be utilized across an expansive scope of innovation stages, including medical device stages, business "off-the-rack" stages, and virtual organizamples. Such programming was recently alluded to by industry, worldwide controllers, and medical services suppliers as "independent programming," "medical device programming," as well as "wellbeing programming," and can once in a while be mistaken for different kinds of programming.
Tips to find investors for SaMD startup
Layout the Business Case
An imperative guideline to remember all through the gathering pledges process is to keep it straightforward. Since quite a while ago, the The field-tested strategy and go-to-showcase technique shouldn't be a million p ago drafted with fantastic detail. All things considered, place emphasis on strong statistical surveying and evidence of ideas.
Make sure to begin the interaction with exhaustive statistical surveying posing the six inquiries underneath to kick you off:
➢ For what reason is this medical device fundamental?
➢ Does this medical device have a demonstrated clinical need?
➢ Are there some other existing devices like mine?
➢ Who is the end client of the medical device?
➢ Who will buy this clinical device?
➢ What is the market size for the device?
Know Your Investor and Risk Scenarios
It's a well-known investors fact that hazard is at the bleeding edge of virtually every financial investor's psyche. Contributing is tied in with bringing in cash without taking such a large number of grand risks. Be that as it may, in medical device subsidizing, financial investors are worried about three kinds of hazard specifically:
➢ Specialized Risk: will the thought work?
➢ Business hazard: does the gadget have a market with a need?
➢ Individuals hazard: are the perfect individuals on staff to make it work long haul?
Being acquainted with these kinds of hazards and knowing what financial investors are searching for will assist organizations with boosting their opportunities for progress.
For instance, a few financial investor's search for little danger, while others acknowledge high risk for high likely rewards. Knowing what your ideal fit would seem like will make drawing in them a lot simpler.
Evidence of Concept prototype
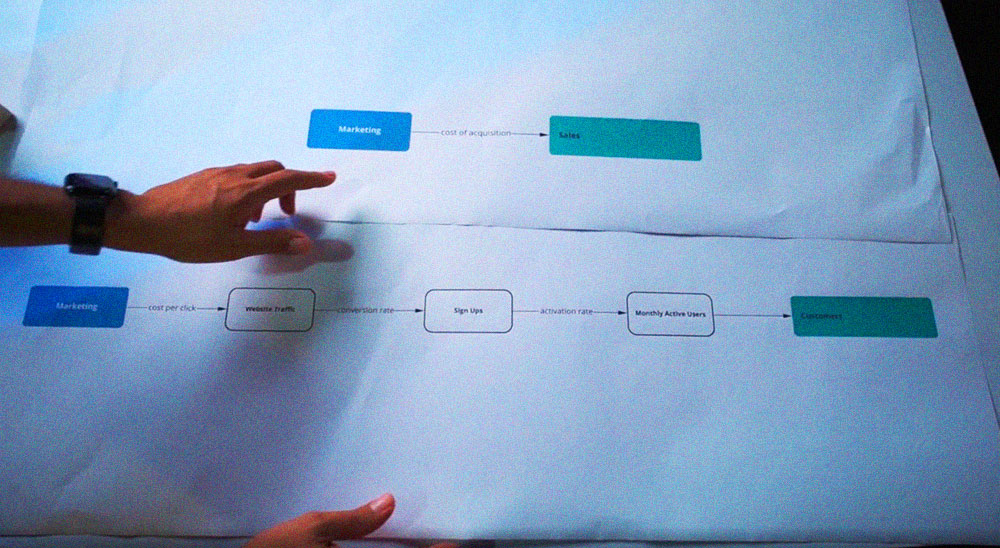
To move your medical device from a plan to a demonstrated idea that will gather consideration from financial investors, you ought to think about building a model. It doesn't need to be exhibited in its last, go-to-showcase state, yet it needs to demonstrate the idea of your gadget.
When done effectively, models can be extremely helpful for conveying the vision of your medical device during item advancement. It can permit outer partners, including financial investors, to more readily comprehend your item’s drawn-out objectives and vision.
Designate Your Resource Wisely
With regards to carrying out a QMS, we emphatically suggest "right-estimating" it with your organization. Right-estimating, your QMS, essentially implies working out your QMS as you go. Contingent upon your business goals, it may not sense well to carry out a completely stacked QMS with every one of the extravagant accessories. All things being equal, you should just execute the key cycles important to satisfy your particular quality and administrative necessities.
The QMS processes most regularly required during item improvement are configuration controls, hazard the board, and report the executives. Contingent upon whether you'll rethink anything, you will require the provider the board processes set up, too.
You probably have restricted assets, so center around the critical cycles vital for the development of your organization. Consider it triaging your cycles by allocating each with levels of significance to the wellbeing of your QMS. In the event that you distinguish that X is crucial to your quality necessities, start there; then, at that point, delay until you have the experimental proof to help carry out Y and Z.
This sort of commonsense methodology will be exceptionally alluring to possible financial investors. By showing your capital productivity and knowledge of your clinical gadget’s cycles and quality requirements, you can set up a significant feeling of trust with financial investors.

Conclusion
What might it take for you as a person to put the blessing on your device? What might make it alright to be utilized by a friend or family member or myself?
Your reaction and position on the matter are significant. How you intend to accomplish the ideal result for your eventual outcome and how achievement will be estimated is basic data to know.
Put yourself in the shoes of a financial investor. What do they have to see to compose the check? You want to show that your innovation merits financing and that your group has the information and abilities to convey what is guaranteed.
If you can give affirmation to a financial investor that your device will satisfy its expected reason to one day be utilized by their friends and family, then, at that point, there's a decent possibility you will actually want to tie down subsidizing your device to get that going.
How to find investors for SaMD startup?
As innovation keeps on propelling all aspects of medical services, programming has turned into a significant piece, all things considered, coordinated generally into advanced stages that serve both clinical and non-clinical purposes. Programming, which all alone is a medical device – Software as a Medical Device – is one of three sorts of programming identified with clinical gadgets. The other two kinds of programming identified with medical devices incorporate programming vital to a clinical gadget (Software in a medical device) and programming utilized in the production or upkeep of a clinical gadget.
What is Software as a Medical Device?
The term Software as a Medical Device External Link Disclaimer is characterized by the International Medical Device Regulators Forum (IMDRF) as "programming planned to be utilized for at least one clinical purpose that plays out these reasons without being important for an equipment medical device."

Utilization of Software as a Medical Device is proceeding to increment. The software as a medical device market is expected to reach $86 billion in 2027. Increasing SaMD popularity resides in the capability to facilitate disease diagnosis, management, and treatment using a SaMD on general-purpose deviTo give some examples, its. It tends to be utilized across an expansive scope of innovation stages, including medical device stages, business "off-the-rack" stages, and virtual organizamples. Such programming was recently alluded to by industry, worldwide controllers, and medical services suppliers as "independent programming," "medical device programming," as well as "wellbeing programming," and can once in a while be mistaken for different kinds of programming.
Tips to find investors for SaMD startup
Layout the Business Case
An imperative guideline to remember all through the gathering pledges process is to keep it straightforward. Since quite a while ago, the The field-tested strategy and go-to-showcase technique shouldn't be a million p ago drafted with fantastic detail. All things considered, place emphasis on strong statistical surveying and evidence of ideas.
Make sure to begin the interaction with exhaustive statistical surveying posing the six inquiries underneath to kick you off:
➢ For what reason is this medical device fundamental?
➢ Does this medical device have a demonstrated clinical need?
➢ Are there some other existing devices like mine?
➢ Who is the end client of the medical device?
➢ Who will buy this clinical device?
➢ What is the market size for the device?
Know Your Investor and Risk Scenarios
It's a well-known investors fact that hazard is at the bleeding edge of virtually every financial investor's psyche. Contributing is tied in with bringing in cash without taking such a large number of grand risks. Be that as it may, in medical device subsidizing, financial investors are worried about three kinds of hazard specifically:
➢ Specialized Risk: will the thought work?
➢ Business hazard: does the gadget have a market with a need?
➢ Individuals hazard: are the perfect individuals on staff to make it work long haul?
Being acquainted with these kinds of hazards and knowing what financial investors are searching for will assist organizations with boosting their opportunities for progress.
For instance, a few financial investor's search for little danger, while others acknowledge high risk for high likely rewards. Knowing what your ideal fit would seem like will make drawing in them a lot simpler.
Evidence of Concept prototype
To move your medical device from a plan to a demonstrated idea that will gather consideration from financial investors, you ought to think about building a model. It doesn't need to be exhibited in its last, go-to-showcase state, yet it needs to demonstrate the idea of your gadget.
When done effectively, models can be extremely helpful for conveying the vision of your medical device during item advancement. It can permit outer partners, including financial investors, to more readily comprehend your item’s drawn-out objectives and vision.
Designate Your Resource Wisely
With regards to carrying out a QMS, we emphatically suggest "right-estimating" it with your organization. Right-estimating, your QMS, essentially implies working out your QMS as you go. Contingent upon your business goals, it may not sense well to carry out a completely stacked QMS with every one of the extravagant accessories. All things being equal, you should just execute the key cycles important to satisfy your particular quality and administrative necessities.
The QMS processes most regularly required during item improvement are configuration controls, hazard the board, and report the executives. Contingent upon whether you'll rethink anything, you will require the provider the board processes set up, too.
You probably have restricted assets, so center around the critical cycles vital for the development of your organization. Consider it triaging your cycles by allocating each with levels of significance to the wellbeing of your QMS. In the event that you distinguish that X is crucial to your quality necessities, start there; then, at that point, delay until you have the experimental proof to help carry out Y and Z.
This sort of commonsense methodology will be exceptionally alluring to possible financial investors. By showing your capital productivity and knowledge of your clinical gadget’s cycles and quality requirements, you can set up a significant feeling of trust with financial investors.

Conclusion
What might it take for you as a person to put the blessing on your device? What might make it alright to be utilized by a friend or family member or myself?
Your reaction and position on the matter are significant. How you intend to accomplish the ideal result for your eventual outcome and how achievement will be estimated is basic data to know.
Put yourself in the shoes of a financial investor. What do they have to see to compose the check? You want to show that your innovation merits financing and that your group has the information and abilities to convey what is guaranteed.
If you can give affirmation to a financial investor that your device will satisfy its expected reason to one day be utilized by their friends and family, then, at that point, there's a decent possibility you will actually want to tie down subsidizing your device to get that going.